Abstract
Background: Immune aplastic anemia (AA) results from cytotoxic T-lymphocyte-mediated destruction of hematopoietic stem cells. Although upfront immunosuppressive therapy (IST) is effective, patients, with relapsed and refractory disease need further treatment with a second round of IST or bone marrow transplantation (BMT). In this retrospective study, we assess the safety and efficacy of alemtuzumab combined with eltrombopag (EPAG) as second-line IST.
Methods: Severe AA patients who relapsed after or were refractory to IST (horse anti-thymocyte globulin, cyclosporine and EPAG) were treated with alemtuzumab and EPAG in combination at the Clinical Center between 2017-2022. Alemtuzumab was given as previously reported in SAA: 10 mg/dose/day for 10 days as an intravenous infusion after a 1 mg test dose in adults and in children < 50Kg, 0.2mg/kg/day for 10 days (not to exceed 10 mg/day). EPAG was administered at 150 mg daily in adults and weight based for children as previously reported. Periodic blood counts and bone marrow evaluations were performed to assess for response and progression to myeloid malignancy. Toxicity was assessed based on the previous profile of the two drugs.
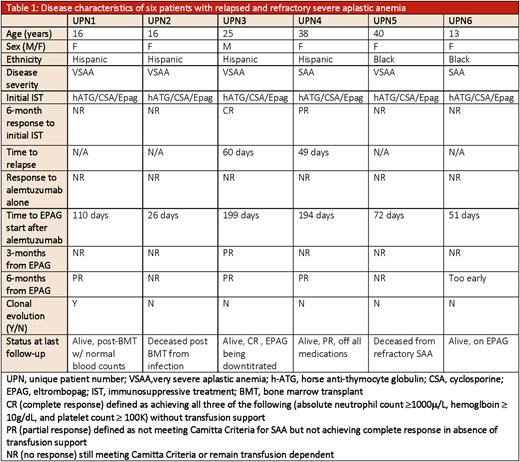
Results: Six patients were included with a median age of 16 years (range 13- 39 years); half were children. Four of six had very severe disease at SAA presentation and the majority were refractory to initial IST. Two patients with relapsed SAA had achieved a response to IST at 6-month evaluation but then had declining blood counts at 60 and 49 days, respectively.
All patients received alemtuzumab as second-line IST (Table 1). EPAG was subsequently added at a median of 60 days from alemtuzumab administration (range 26 days - 199 days) when there was no signs of response or when the counts declined further, .was performed at 3-months and 6-months from EPAG addition. Both patients with relapsed disease achieved a response at 6-month evaluationUPN3 showed early partial response (PR) at 3-months and then ultimately achieved a complete response (CR); at last follow-up, EPAG was being down-titrated with stable blood counts and no signs of evolution to myeloid malignancy. UPN4 achieved a PR at 6 months and at last follow-up maintained stable counts despite discontinuation of EPAG.
Of the four refractory patients, only one (UPN1), responded to the addition of EPAG and continued treatment for 2 years. While the EPAG was reduced in dosage, evaluation for declining blood counts with a bone marrow biopsy revealed progression to acute myeloid leukemia, with monosomy 7 and an ASXL1 clone at 24% variant allele frequency. The patient achieved normal blood counts post BMT. Two of the non-responders (UPN2 and UPN5) died from SAA complications; UPN2 died shortly after BMT from refractory infection. UPN6 also had refractory disease; she is 5 months from alemtuzumab and 3 months from EPAG treatment without a response undergoing evaluation for BMT.
Patients that responded (UPN1, UPN3, and UPN4) had a median hemoglobin baseline of 7 g/DL (range 6-7.6 g/DL) and increased to a median of 9.3 g/DL (range 8.4-11.6 g/DL), 6 months after EPAG. Responders had a median ANC baseline of 0.69 k/mcL (range 0.46-1.58 k/mcL) and increased to a median of 1.5 k/mcL(range 1.5-2.11). At baseline they also presented with a median platelet of 17 k/mcL at baseline (range 5-18 k/mcL) and increased to 23 k/mcL (range 23-37 k/mcL), 6 month,s after EPAG.
None of the patients had toxicity, specifically liver function abnormalities, with the addition of EPAG. The median AST and ALT were 36 U/L (range 20-66 U/L) and 93 U/L (range 13-170 U/L) 6 months after EPAG, respectively. Patients had a median total bilirubin of 0.4 mg/DL (range 0.3-0.9 mg/DL).
Conclusion: In absence of BMT, second-line IST is administered for relapsed and refractory SAA patients. The addition of EPAG to alemtuzumab after 1-3 months resulted in responses in relapsed patients but not in refractory cases. The combination was safe without increased toxicities, but prolonged use of EPAG in a single refractory patient associated with development of myeloid malignancy.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.